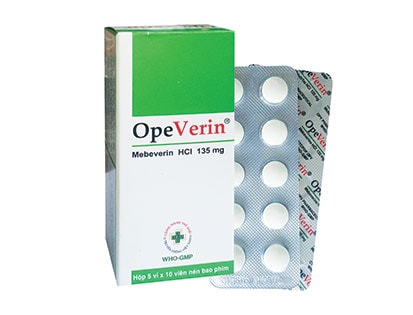
THÀNH PHẦN:
Mỗi viên nén bao phim chứa:
- Hoạt chất: Mebeverin HCl 135mg
- Tá dược: Lactose monohydrat, cellulose vi tinh thể, natri starch glycolat, povidon, talc, magnesi stearat, opadry II white.
MÔ TẢ SẢN PHẨM:
Viên nén tròn bao phim, màu trắng ngà.
DƯỢC LỰC HỌC:
Mebeverin là một thuốc trị co thắt, có tác động trực tiếp lên cơ trơn của ống tiêu hóa.
DƯỢC ĐỘNG HỌC:
Mebeverin được hấp thu nhanh sau khi uống nồng đổ đỉnh trong huyết tương đạt được trong khoảng 1-3 giờ. 75% mebeverin gắn kết với albumin trong huyết tương. Mebeverin bị chuyển hóa hoàn toàn thành acid veratric và mebeverin alcohol. Các chất chuyển hóa bị thải trừ qua đường nước tiểu.
CHỈ ĐỊNH:
Điều trị hội chứng ruột kích thích và các tình trạng: kích thích đại tràng mãn tính, táo bón do co thắt, viêm niêm mạc đại tràng, viêm đại tràng co thắt với các triệu chứng như đau quặn bụng, tiêu chảy không đặc hiệu dai dẳng (kèm hoặc không kèm với táo bón xen kẽ) và đầy bụng.
CÁCH DÙNG-LIỀU DÙNG:
Người lớn và trẻ em trên 10 tuổi: 1 viên x 3 lần/ ngày, uống 20 phút trước bữa ăn.
Nuốt viên thuốc với nước, không nhai thuốc.
CHỐNG CHỈ ĐỊNH:
Quá mẫn với bất kỳ thành phần nào của thuốc.
LƯU Ý VÀ THẬN TRỌNG:
Sử dụng ở trẻ em: mebeverin không được khuyến cáo dùng cho trẻ em dưới 10 tuổi.
Phụ nữ có thai và cho con bú: Không khuyến cáo sử dụng mebeverin cho phụ nữ có thai và đang cho con bú.
Tác động của thuốc khi lái xe và vận hành máy móc: Chưa có nghiên cứu về tác động của thuốc khi lái xe và vận hành máy móc.
TÁC DỤNG KHÔNG MONG MUỐN:
Nói chung, mebeverin dung nạp tốt. Vài tác dụng phụ như phát ban ở da, mày đay và phù mạch có thể xảy ra.
Thông báo cho bác sĩ những tác dụng không mong muốn gặp phải khi sử dụng thuốc.
TƯƠNG TÁC THUỐC:
Chưa có báo cáo về tương tác thuốc.
QUÁ LIỀU VÀ CÁCH XỬ TRÍ:
Triệu chứng: Về lý thuyết, thuốc có thể gây kích thích hệ thần kinh trung ương khi sử dụng quá liều mebeverin. Trong trường hợp này, các triệu chứng thường không có hoặc ở mức độ nhẹ và nhanh chóng tiêu biến.
Xử trí: Chưa có biện pháp xử trí được khuyến cáo.
TRÌNH BÀY:
Hộp 5 vỉ x 10 viên nén bao phim.
BẢO QUẢN: Ở nhiệt độ dưới 300C, nơi khô ráo, tránh ánh sáng.
HẠN DÙNG: 36 tháng kể từ ngày sản xuất. Không dùng thuốc khi quá hạn sử dụng.